|
1/13/2024 0 Comments Atomic mass of carbonWeight is a measure of force affected by Earth's gravitational pull. (Of course it would make no more sense to measure atoms in pounds or kilograms than to measure the width of a hair in light-years but pounds and kilograms are the most familiar units of weight and mass respectively.) Though the two are relatively convertible on Earth, they are actually quite different. However, the pound is a unit of weight in the English system, whereas a kilogram is a unit of mass in the metric and SI systems. Mass is, after all, basically the same as weight, is it not? In fact it is not, though people are accustomed to thinking in those terms since most weight scales provide measurements in both pounds and kilograms. 
One might ask why such pains have been taken to make the distinction. Though "weigh" is used as a verb in this essay, this is only because it is less cumbersome than "measure the mass of." (In addition, "atomic weight" may be mentioned when discussing studies by scientists of the nineteenth century, who applied that term rather than atomic mass.) Indeed, the use of " atomic weight" today merely reflects the fact that scientists in the past used that expression and spoke of "weighing" atoms. The first of these is not as accurate as the second, which explains why atomic mass was chosen as the subject of this essay. Some textbooks and other sources use the term atomic weight instead of atomic mass. The measurement of atomic mass was thus a historic challenge that had to be overcome, and the story of the ways that scientists met this challenge is an intriguing one. More specifically, the work of a chemist requires the use of accurate atomic proportions in forming the molecules that make up a compound. One answer is that everything is made of atoms. But what about something so insignificant in mass that comparing it to a gram is like comparing a millimeter to the distance between Earth and the nearest galaxy? Obviously, special units are needed for such measurements then again, one might ask why it is necessary to weigh atoms at all. Geological specimens are known in which the element has an isotopic composition outside the limits for normal material.Every known item of matter in the universe has some amount of mass, even if it is very small.See table 1 for details of range and original paper for the atomic weight of the element from different sources.However three such elements (Th, Pa, and U) do have a characteristic terrestrial isotopic composition, and for these an atomic weight is tabulated. , indicates the mass number of the longest-lived isotope of the element. Substantial deviations in atomic weight of the element from that given in the Table can occur. Modified isotopic compositions may be found in commercially available material because it has been subject to an undisclosed or inadvertant isotopic fractionation.Value being given the tabulated value should be applicable to any normal material. Range in isotopic composition of normal terrestrial material prevents a more precise.The difference between the atomic weight of the element in such specimens and that given in the Table may exceed the stated uncertainty. Geological specimens are known in which the element has an isotopic composition outside the limits for normal material.See original paper for the range of these elements from different sources 
List of Elements with Range of Atomic Weights. 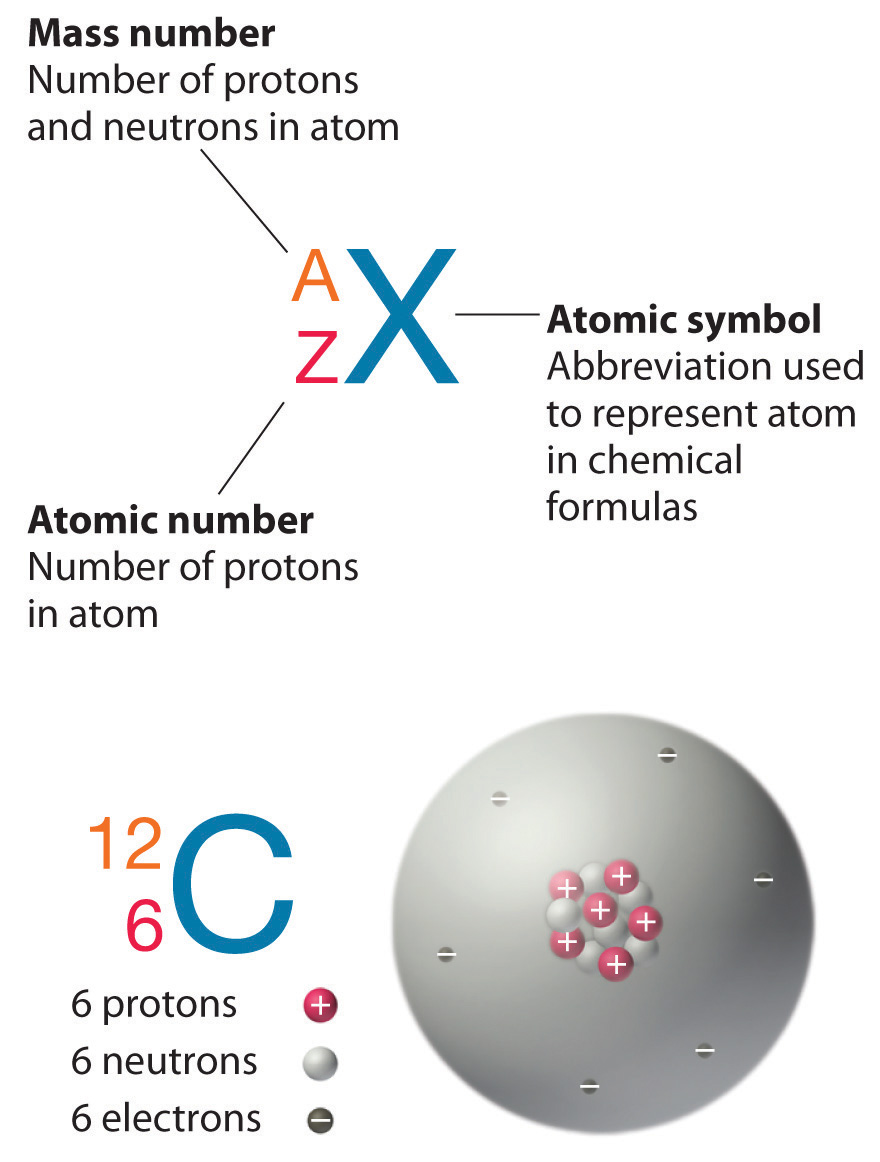
See also a copy of the periodic table with atomic weights to five significant figures. See below for the elements listed in Atomic Number Order or Name order. The original paper should be consulted for full details of the variation in atomic weight and the half life of the radioisotopes quoted below.Ī number in parentheses indicates the uncertainty in the last digit of the atomic weight. For radioactive elements the isotope with the longest half-life is quoted in parenthesis. In the other lists the values quoted are those suggested for material where the origin of the sample is unknown. The standard atomic weights of twelve elements having two or more stable isotopes have variability of atomic-weight values in natural terrestrial materials. Previous values may be consulted from the 1993 table, the 1995 table, the 1997 table, the 1999 table, the 2001 table, the 2005 table, the 2007 table, the 2009 table, the 2011 table, the 2013 table, the 2015 table or the 2019 table. World Wide Web version of atomic weight data originally prepared by G. These tables are based on the 2021 table with changes from the 2019 table for the values of Ar, Hf, Ir, Pb and Yb and changes to the uncertainty for Al, Au, Co, F, Ho, Mn, Nb, Pa, Pr, Rh, Sc, Tb, Tm, and Y. 2021 Atomic Weights IUPAC Commission on Isotopic Abundances and Atomic Weights.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
